Hartford Courant
CDC Panel Includes Transportation Workers on 'Essential' List for Vaccine Priority
[Ensure you have all the info you need in these unprecedented times. Subscribe now.]
A federal advisory panel is recommending that the next round of coronavirus vaccines be given to teachers, first responders, correction officers and grocery store employees, along with people 75 and over.
The vaccine is currently being given to group 1a, health care workers and residents and staff at long-term care facilities. Group 1b will include “frontline essential workers.” Truck drivers and other transportation workers fall into Group 1c as “essential workers.” They are only recommendations, made by Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention, but many states are expected to follow them.
But even as the ACIP made its recommendations Dec. 20 on who should get the vaccine next, officials acknowledged that there still isn’t enough vaccine available for people who make up the second and third round of vaccinations.
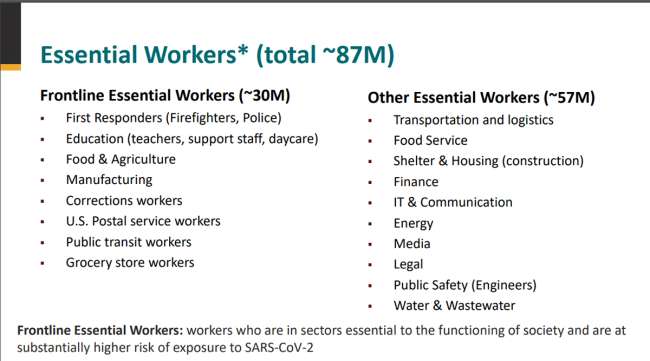
There are 51 million people in the next round, or 1b category: an estimated 30 million frontline essential workers and another 21 million age 75 and over.
Federal officials say there is enough vaccine to inoculate about 20 million people in December, another 30 million in January, another 50 million in February for a total of 100 million doses total. But since the third phase, or 1c category, includes about 129 million people, there will not be enough vaccine to handle all of the people targeted for the next two rounds, at least by the end of February.
Several members of the 14-member commission raised questions about the CDC’s recommendations about why they were separating out 65-74 year-olds and why some work groups were considered “frontline essential” workers as opposed to just “essential” workers.

A health care worker injects a vaccine into a patient's arm. (Andrey Rudakov/Bloomberg News)
At one point during the hearing, Chairman Dr. Jose Romero defended the ACIP’s review process against what he said were allegations that they were excluding some racial groups.
“It is important that the public understand throughout our deliberations and careful evaluation of the data the ACIP has struggled painfully to deal with distribution of a limited resource of vaccine,” Romero said. “Our attempt has been always to achieve an equitable, ethical and fair distribution of that resource.”
The CDC defines frontline essential workers as all first responders not included in first phase, teachers, day care workers, prison employees, food and agricultural workers such as at meat processing plants, manufacturing workers, public transit employees, postal employees and grocery store employees.
In the third phase, or 1c category, there are 129 million people that can be vaccinated. This category includes anyone 65-74 years old, anyone under 65 with pre-existing conditions and the remainder of what they are calling “essential workers.”
The group of “essential workers” in category 1c includes food service and restaurant workers, construction workers, transportation employees, legal, media, finance, energy workers and water and wastewater employees.
The first round of vaccinations, which began last week, were clear on who was going to vaccinated — hospital workers and nursing home residents — but this time there is leeway for each state to decide who gets vaccinated next.
Vaccinations of the first groups started on Dec. 14. It is expected the next group will be getting shots made by Pfizer-BioNTech or Moderna. The FDA approved the Moderna vaccine for emergency use on Dec. 19. About 556,000 Americans have been vaccinated thus far, according to the CDC.
The CDC on Dec. 20 also released some early data on reactions to the vaccinations.
As of Dec. 18, scientists said there had been six cases of anaphylaxis shock among the first 112,807 people to get the Pfizer vaccine either in Great Britain or the United States.
Overall there have 3,150 “health care events” among that first group to vaccinated. The CDC defines a health care event as being unable to perform normal daily activities, unable to work or required care from a doctor or health care professional.
The national rollout to vaccinate nursing home residents and staff actually started late last week with a “soft” opening in facilities in Connecticut, Florida, Ohio and West Virginia.
The federal government through Operation Warp Speed made a deal with CVS and Walgreens to administer the vaccine to all nursing home residents and staff in the country.
They must schedule three clinics at each facility to ensure that everyone gets an opportunity to get two shots. Both the Pfizer and Moderna vaccines require a second booster shot anywhere from two to four weeks after the first one.
Want more news? Listen to today's daily briefing:
Subscribe: Apple Podcasts | Spotify | Amazon Alexa | Google Assistant | More
Distributed by Tribune Content Agency, LLC




